Healthcare Innovation Procurement Resource Hub
Pre-Commercial Procurement (PCP) and Public Procurement of Innovation (PPI) are strategic instruments that enable public authorities to stimulate the development and adoption of innovative solutions. In the healthcare sector, these tools are particularly relevant, as they support the acquisition of innovative products and services that transform the way healthcare is delivered, improving efficiency, sustainability, quality of care, and patient outcomes.
There are tons of information available on innovation procurement. This repository groups all key information and resources related to PCP and PPI in healthcare.
What will you find in this page?
What is Pre-commercial Procurement (PCP) in Healthcare?
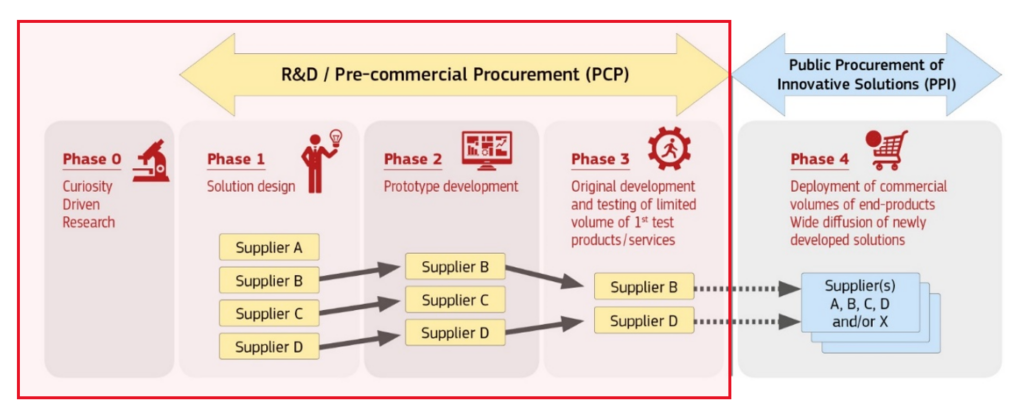
Pre-commercial procurement (PCP) is a procurement procedure where public procurers buy research and development services from several suppliers competing in parallel to compare alternative solution approaches and identify the best value for money solutions that the market can deliver to address their needs. Research and development are split into phases (solution design, prototyping, original development and validation/testing of a limited set of first products) with the number of competing research and development providers being reduced after each phase.
The public buyer does not reserve the R&D results for its own use, but shares them with the economic operators under market conditions. The risks and benefits of the R&D are thus shared between the public buyer and the industry.
PCP does not entail the purchase of a finished product!
Why use a PCP?
The benefits of conducting a PCP as a public buyer:
- Ability to address complex and unmet needs;
- Closer contact with the market players and suppliers;
- Sharing of risks and benefits between public buyer and suppliers;
- Leads to better value for money in later procurement procedures, as you can filter out technological R&D risks of potential alternative solutions before committing to procuring a large scale commercial roll-out.
The benefits of participating in a PCP as a supplier:
- Access to funded R&D: you do not have to carry the full financial risk yourself;
- Early validation of your technology;
- Faster route to the market;
- Reduced commercial risk, as there is no obligation to deliver a finished product, only to carry out R&D activities.
When is a PCP appropriate?
When there is no suitable innovative solution on the market to address your unmet need, meaning that significant R&D is required.
PCP Process and Phases
- PHASE 1: Solution design
- PHASE 2: Prototype development
- PHASE 3: Pilot testing and validation
During each of the PCP Phases, the performance of each participating provider will be monitored and evaluated against pre-defined criteria. After each Phase, the best scoring offers will be awarded a contract for the next Phase. The end of Phase 3 marks the end of the PCP procedure.
Key success factors
- Clear definition of your unmet need;
- Clear and outcome-oriented demand;
- Strong patient involvement;
- Open engagement with the market;
- Well-designed evaluation criteria.
Legal framework
PCP is exempt from the EU Public Procurement Directives
❗PCPs must comply with the fundamental EU principles of transparency, equal treatment, non-discrimination and proportionality.
What is Public Procurement of Innovation (PPI) in Healthcare?
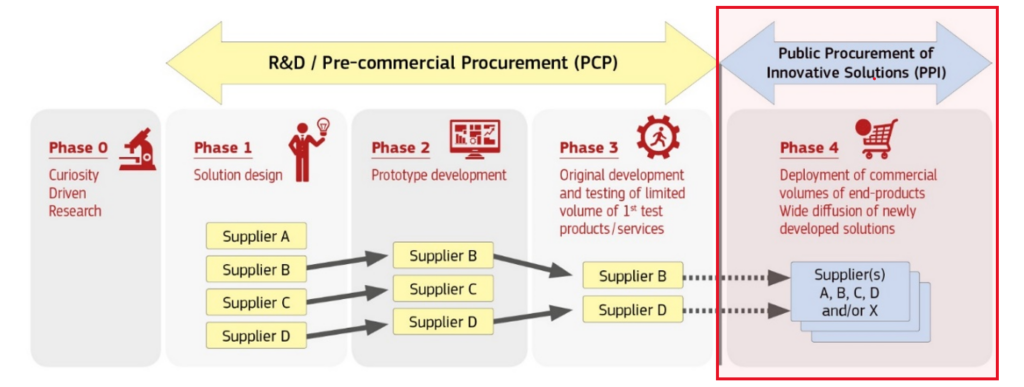
Public procurement of innovative solutions (PPI) is a procurement approach where the public sector uses its purchasing power to act as early adopter of innovative solutions which are not yet available on large scale commercial basis. It provides a large enough demand to incentivize the industry to invest in wide commercialisation to bring innovative solutions to the market with the quality and price needed for mass market deployment. This enables the public sector to modernise public services with better value for money solutions and provides growth opportunities for companies.
Why use a PPI?
Benefits of conducting a PPI as a public buyer:
- Better value for money;
- Access to innovative solutions that address arising needs with improved quality, efficiency and user experience;
- Creates a clear demand for innovation, encouraging the industry to invest and scale innovative solutions.
Benefits of participating in a PPI as a supplier:
- The public buyer serves as a first customer, providing early market traction;
- Deploy and validate the solution at operational scale in real-life setting;
- Facilitates scaling and repeatability, as public demand helps standardize solutions and opens opportunities for wider market uptake;
- Credibility boost, supporting further commercialization and entry into new markets.
When is a PPI appropriate?
When innovative solutions already exist but are not yet widely adopted or commercialised, meaning that no new R&D is needed.
PPI Process
Preparation: Define your unmet need, engage with the market and build a business case.
Procurement: Choose the appropriate procedure (open, restricted, competitive procedure with negotiation, competitive dialogue or innovation partnership) as best-suited for the innovative solution desired. More flexible and dialogue-based procedures allow closer interaction with suppliers, helping to refine requirements, adapt solutions and reduce implementation risks when procuring innovative solutions.
Deployment: Execute the contract and monitor the performance of the contractor.
Key success factors
- Clear definition of your unmet need;
- Clear and outcome-oriented demand;
- Strong patient involvement;
- Open engagement with the market.
Legal framework
PPIs must follow EU public procurement rules and corresponding national implementation provisions.
Most important are the 2014 Public Procurement Directives.
- Public sector: Directive 2014/24/EU of the European Parliament and of the Council of 26 February 2014 on public procurement and repealing Directive 2004/18/EC Text with EEA relevance
- Utilities: Directive – 2014/25 – EN – EUR-Lex; EUR-Lex – 02014L0025-20260101 – EN – EUR-Lex
These Directives establish the legal framework governing public procurement across the EU, including provisions that enable innovation-friendly procedures.
They introduce instruments such as the Innovation Partnership and clarify flexibility for functional specifications and competitive procedures.
Public hospitals and health authorities are considered contracting authorities under this Directive. It governs procurement of:
- Medical technologies
- Digital health solutions
- Surgical, medical and diagnostic procedures
- Pharmaceuticals (with specific considerations)
- Health services contracts
- Innovation partnerships
- PCP/PPI-related procedures (when applicable)
It includes provisions enabling innovation-friendly tools such as:
- Innovation Partnership (Article 31)
- Competitive Dialogue
- Competitive Procedure with Negotiation
- Functional/Performance-based specifications
Pilot vs. Innovation Partnership vs. PCP/PPI: Key Differences Explained
The differences between Innovation Procurement (PCP/PPI), Innovation Partnership, and Pilots are not always clear in practice, but they differ substantially in ambition, legal framework, scalability, and market-shaping capacity. The table below provides a structured comparison to help decision-makers and procurers identify which approach best fits their objectives, the maturity of the solution, and the level of transformation they aim to achieve.
|
Dimension |
Pilot |
Innovation Partnership |
Innovation Procurement (PCP / PPI) |
|
Objective |
Test / validate a specific solution |
Develop and purchase innovative solution within one procedure |
Develop or deploy solutions not fully available on the market |
|
Length / Difficulty |
Relatively short & lighter |
Medium-High complexity |
High complexity, multi-phase |
|
Market Influence Power |
Limited, adopts existing or near-ready solution |
Influences specific supplier development |
Shapes the market; stimulates competition; creates future supply |
|
Competition Level |
Often single supplier |
Competitive selection → then single partner(s) |
Competitive phased process with multiple suppliers |
|
Risk Distribution |
Higher dependency on one supplier |
Shared between buyer and selected partner |
Risk shared across multiple suppliers |
|
Scalability Potential |
Scaling uncertain / not embedded structurally |
Scalable within contracting authority |
Designed for replication & broader adoption → systemic transformation |
|
When NOT to Use |
If systemic change or market shaping is required |
If need is exploratory or unclear |
If solution already fully available and standard procurement suffices |
Complete Repository of EU Healthcare PCP and PPI Projects
| Project Name | Instrument | Health Topic | Participating Countries |
|---|---|---|---|
| Nightingale | PCP | Patient monitoring (wearable, clinical) | Netherlands, Germany, Sweden, Belgium, UK |
| ANTISUPERBUGS | PCP | Infection control, antimicrobial detection | UK, Germany, Italy, Spain |
| DECIPHER | PCP | Electronic Health Records, patient monitoring | Spain, UK, Italy |
| HSMONITOR | PCP | Remote health monitoring | Turkey, Sweden, Croatia, Italy |
| PROEMPOWER | PCP | Diabetes management solution | Turkey, Portugal, Spain, Bulgaria, Italy |
| TIQUE | PCP | Advance Heart Failure Integrated Care | Spain, Sweden, Italy, UK, Israel |
| CRANE | PCP | Chronic patients integrated self-management | Norway, Sweden, Spain, Denmark |
| Instand-NGS4P | PCP | Cancer NGS workflow standardisation | Austria, Germany, Italy, Netherlands, UK, Slovenia |
| oncNGS | PCP | Oncology genomic diagnostics | Belgium, Germany, Italy, Spain, France |
| eCARE | PCP | Frailty & emergency care | Spain, Italy, Germany, Portugal |
| NYMPHA | PCP | Bipolar disorder management | Italy, Denmark, Spain |
| STOPandGO | PPI | Telecare and telehealth for older people | UK, Spain, Netherlands, Italy |
| RITMOCORE | PPI | Arrhythmia remote monitoring | Spain, UK |
| HAPPI | PPI | Healthy ageing / assistive tech | Ireland, Belgium, Italy, Greece, Germany, Spain, Portugal, Cyprus, France, Finland, Czechia, Sweden, UK, Norway |
| SILVER | PCP | Elderly independent living robotics | UK, Denmark, Finland, Netherlands, Sweden |
| RaDAR | PPI | Rapid detection of antimicrobial resistance | Spain, France, Italy |
| Thalea & Thalea II | PCP & PPI | Telemedicine for co-morbid patients | Germany, Netherlands, Spain, Belgium, Finland |
Guidelines for Healthcare Innovation Procurement (PCP/PPI)
European-level guidance provides the legal, strategic, and methodological foundation for implementing PCP and PPI in any domain field (environmental science, energy, transportation, urban mobility, healthcare….).
- European Commission Guidance on Innovation Procurement (2021/C 267/01)
- Provides practical, non-binding guidance to help public procurers to buy innovative solutions, rather than just off-the-shelf products
- Guidance documents on PCP and PPI implementation under Horizon 2020 and Horizon Europe
- PCP Guidance (Horizon 2020)
- PPI Guidance (Horizon 2020)
- Horizon Europe innovation Procurement
- Public Procurement Directives.
- European Assistance for Innovation Procurement (EAFIP)
- Provides technical assistance, training, and case studies to support contracting authorities in implementing innovation procurement. Although the support is not focused on innovation procurement in the healthcare sector specifically, it offers practical examples, templates, and lessons learned from real PCP and PPI projects that were carried out across Europe.
- Health Innovation Procurement Adoption Tool (HIPAT)
- Step-by-step guide on public procurement of innovation specific to healthcare. It includes curated resources and ad-hoc support services.
- Innovation Procurement Platform
- Repository of resources (sector-agnostic)
- ProcurCompEU tool (OECD powered)
- Tool for public procurement professional to identify missing competences or areas where further capability-building initiatives could be implemented and lessons learned .
- Adaptation of ProcurCompEU for innovation in Healthcare procurement.
Funding Opportunities for Healthcare PCP and PPI Projects
EU-level funding and project databases:
- EU Funding & Tenders Portal – Central portal for all EU funding programmes https://ec.europa.eu/info/funding-tenders/opportunities/portal
- TED (Tenders Electronic Daily) – Official EU procurement portal; browse PPI/PCP calls and ongoing tenders. https://ted.europa.eu/
- CORDIS – Database of EU-funded projects; identify existing PCP/PPI projects and potential partners. https://cordis.europa.eu/
- EU4Health Programme – Health-system resilience and innovation actions. https://health.ec.europa.eu/funding/eu4health_en
- Interreg Europe – Regional cooperation funding for innovation pilots. https://www.interregeurope.eu/
- InvestEU – Financial instruments for innovation and infrastructure via EIB. https://europa.eu/investeu/home_en
- Horizon Europe funding for PCP and PPI – Research and innovation
- EIC Innovation Procurement Programme – European Innovation Council
Innovation Procurement Communities and Networks in Europe
- Competence Centers on Innovation Procurement: National organisations with the mandate to support the use of PCP/PPI (practical and/or financial assistance to public procurers in the preparation and/or implementation of PCP and PPI across all sectors of public interest).
- Public Buyers Community: European Commission initiative dedicated to facilitating joint action in public purchasing across the EU.
- Innovation Procurement Hubs (IPH) – Dedicated Community of Practice (CoP)
- Health Proc Europe: Non-profit interest group of European and Canadian hospital and health care procurement managers aiming to create the largest digital procurement ecosystem.
- Procure4Health: Open community that encourages EU-wide adoption of innovation procurement.
- Agora InnoHSupport: Interactive space designed to connect public procurers, suppliers, and investors in healthcare innovation procurement.
- ECLIPSE | EuroQuity: Connects public procurers with innovators, investors, and other relevant stakeholders. Ecosystem specially focused on investors.
Notice that inactive forums and communities are not shown.
Navigating Innovation Procurement with Confidence: Guidance, Resources and Advisory Support
Understanding innovation procurement is the first step, implementing it is the real challenge. While this page brings together key knowledge and EU healthcare PCP/PPI projects, the Health Innovation Procurement Adoption Tool (HIPAT) helps you move from information to action. Through structured guidance, practical templates, and advisory support, HIPAT supports healthcare procurers in designing and launching successful PCP and PPI processes with clarity and confidence.
Ready to start your innovation procurement journey? Explore HIPAT and access the tools that will guide you step by step.